Surface deep oxidation of ofloxacin and 2,4-dichlorophenol over ferrocene@sepiolite due to their synergistic effect in visible light driven heterogeneous Fenton reaction process†
Abstract
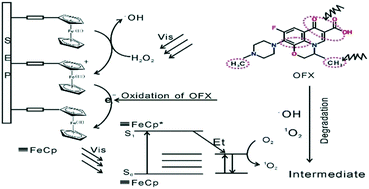
The performance of heterogeneous Fenton-like catalysts was greatly impeded by the aggregation of iron species and release of Fe ions. Herein, we report a useful strategy involving anchoring ferrocene on sepiolite (FeCp@Sep) with high dispersity by –C2H4– chemical bond. Ofloxacin (OFX, 100 ml 10 mg L−1) was 100% degraded in FeCp@Sep system with H2O2 (2.0 mmol L−1) under visible light irradiation (>420 nm). However, only a structural transformation of OFX occurred in FeCp system under the same conditions. The open mesoporous system in FeCp@Sep facilitated the molecular diffusion and transportation. Hence, high concentration of OFX resulted in high removal efficiency in FeCp@Sep system due to the surface profusion of OFX. Moreover, uniformly dispersed and spatially separated FeCp encapsulates Sep, which can not only enhance the reusability of FeCp, but also favor the occurrence of the surface deep oxidation process. Based on the results of ESR analysis and UV-vis spectroscopy, the Fenton degradation process by ˙OH, 1O2, and HO2˙ occurred on the surface of FeCp@Sep instead in the bulk solution. In addition to ˙OH and HO2˙, the involvement of 1O2 was first identified in the Fenton reaction process and their effect on OFX degradation was investigated by HPLC-MS. These findings are important for the expansion of the heterogeneous Fenton catalyst family and its application in water treatment.


 Please wait while we load your content...
Please wait while we load your content...