Mitigating effect of organic matter on the in vivo toxicity of metal oxide nanoparticles in the marine environment†
Abstract
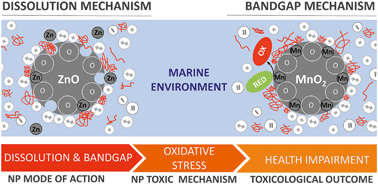
Major constituents of seawater, i.e. ions and natural organic matter (NOM), can influence the environmental and toxicological behaviour of nanoparticles (NPs) in aquatic systems. By adsorbing–ligating–reacting to NP surface reactive sites, they can modify the NP surface structure and overall physico-chemical proprieties. This study explored the fate and in vivo toxicity of ZnO and MnO2 NPs under artificial seawater conditions. These two nanomaterials are representative of metal oxide NPs inducing harm via dissolution and bandgap mechanisms, respectively. To gain a comprehensive understanding of the overall toxicological outcome, we traced the behaviour of NPs in the test systems (i.e. aggregation, sedimentation, dissolution, sorption), their fate in the model organism (i.e. ingestion and cellular internalization by oyster larvae), and the induction of a toxicological pathway (i.e. oxidative stress) up to pathogenesis. We found that ZnO NPs induced harm to oyster larvae under seawater conditions, but NOM mitigated its intensity. In contrast, MnO2 NPs were not toxic at the tested concentrations (up to 200 μM), and their toxicological stasis was not modified by the presence of organic matter. We propose that strong ion sorption on the MnO2 NP surface blocked redox-active sites thus preventing their bandgap mode of action.


 Please wait while we load your content...
Please wait while we load your content...