Engineered superparamagnetic nanomaterials for arsenic(v) and chromium(vi) sorption and separation: quantifying the role of organic surface coatings†
Abstract
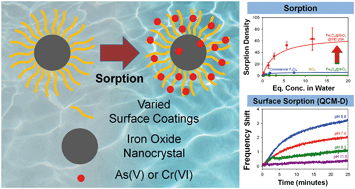
Inorganic–organic nanoscale materials have recently received significant interest as tunable, next generation, sorbents for the separation of metal and metalloid contaminants, including arsenic (As) and chromium (Cr), among others. In this work, we have designed and synthesized IONCs coated with specific functionalized organic materials with the goal of variable explicit evaluation. Specifically, single domain, superparamagnetic, monodisperse IONCs were synthesized and transferred into water via surface functionalization by ligand exchange and encapsulation methods. The as synthesized hybrid materials showed high performance for both As(V) and Cr(VI) sorption when nanocrystals are coated with positively charged organic surface coatings such as polyethyleneimine (PEI) and cetyltrimethylammonium bromide (CTAB). IONC cores coated with negatively charged organic coating materials (polyethyleneglycol (PEG), oleic acid (OA), sodium dodecyl sulfate (SDS)) and silica (SiO2) demonstrated significantly lower sorption capacities. When silica coated IONCs (Fe3O4@SiO2, core–shell materials) were surface coated with PEI, sorption capacities for As(V) and Cr(VI) of Fe3O4@SiO2@PEI are comparable to Fe3O4@PEI, underscoring the importance of surface coating functionality. To complement these studies, real-time sorption behavior of As(V) and Cr(VI) with PEI was explored by quartz crystal microbalance with dissipation (QCM-D).


 Please wait while we load your content...
Please wait while we load your content...