Using chromate to investigate the impact of mineral–organic contact time on the surface reactivity of goethite†
Abstract
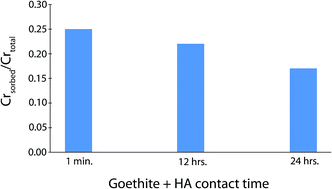
Chromate was used as a chemical probe to investigate the impact of mineral–organic contact time on the surface reactivity of two different sizes of goethite particles. A series of goethite–chromate sorption batch reactions were conducted in the presence and absence of Suwannee River humic acid (HA) and natural organic matter (NOM) using nano- and micro-scale goethite particles. In experiments with added organics the amount of time allowed for goethite–organic matter interaction (i.e. contact time) was varied from less than 1 minute, up to 24 hours prior to the addition of chromate. Results indicated that nano- and micro-scale goethite in the absence of organics sorbed nearly identical amounts of chromate on a per mass basis, despite the greater surface area of the smaller particles. Results also indicated that the presence of ∼10 mg L−1 of HA and a contact time of less than 1 minute reduced the amount of chromate sorbed by both nano- and micro-scale goethite. Increasing the contact time resulted in greater decreases in chromate sorption. Experiments using NOM produced similar results. While chromate sorption was most rapid during the first hour of the experiments, goethite particles continued to sorb additional chromate over a period of up to 7 days. Additionally, a noticeable impact on chromate sorption due to increased contact time was present over that time period.


 Please wait while we load your content...
Please wait while we load your content...