Self-diffusion barriers: possible descriptors for dendrite growth in batteries?
Abstract
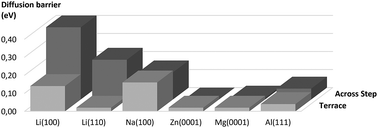
Dendrite formation is one of the most pressing issues in current battery research. Lithium based batteries are prone to forming short-circuit causing dendrites, while magnesium based batteries are not. Recently it was proposed that the tendency towards dendrite growth is related to the height of the self-diffusion barrier with high barriers leading to rough surface growth which that subsequently cause dendrite formation, which was supported by density functional theory calculations for Li, Na and Mg [J. Chem. Phys., 2014, 141, 174710]. We now extend this computational study to zinc and aluminum which are also used as battery anode materials, and we additionally consider diffusion barriers that are relevant for three-dimensional growth such as barriers for diffusion across steps. Our results indicate, in agreement with experimental observations, that Li dendrite growth is an inherent property of the metal, whereas Zn dendrite growth results from the loss of metallic properties in conventional Zn powder electrodes.
- This article is part of the themed collection: 2018 Energy and Environmental Science HOT Articles


 Please wait while we load your content...
Please wait while we load your content...