Enabling alternative ethylene production through its selective adsorption in the metal–organic framework Mn2(m-dobdc)†
Abstract
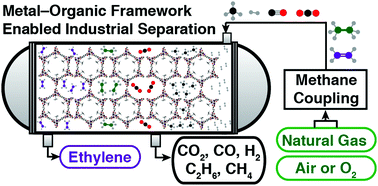
The unique adsorptive properties of metal–organic frameworks open the door to new processes for energy and raw materials production. One such process is the oxidative coupling of methane for the generation of ethylene, which has limited viability due to the high cost of cryogenic distillation. Rather than employing such a traditional separation route, we propose the use of a porous material that is highly selective for ethylene over a wide range of gases in an energy- and cost-effective adsorbent-based separation process. Here, we analyze the metal–organic frameworks M2(m-dobdc) (M = Mg, Mn, Fe, Co, Ni; m-dobdc4− = 4,6-dioxido-1,3-benzenedicarboxylate), featuring a high density of coordinatively-unsaturated M2+ sites, along with the commercial adsorbent zeolite CaX, for their ability to purify ethylene from the effluent of an oxidative coupling of methane process. Our results show that unique metal–adsorbate interactions facilitated by Mn2(m-dobdc) render this material an outstanding adsorbent for the capture of ethylene from the product mixture, enabling this potentially disruptive alternative process for ethylene production.
- This article is part of the themed collection: 2018 Energy and Environmental Science HOT Articles


 Please wait while we load your content...
Please wait while we load your content...