Geometric distortions in nickel (oxy)hydroxide electrocatalysts by redox inactive iron ions†
Abstract
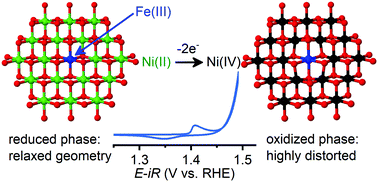
The dramatic change in electrochemical behavior of nickel (oxy)hydroxide films upon incorporation of Fe ions provides an opportunity to establish effective electrocatalyst design principles. We characterize a photochemically deposited series of Fe–Ni (oxy)hydroxides by X-ray absorption spectroscopy and track the voltage- and composition-dependence of structural motifs. We observe a trigonal distortion in di-μ-hydroxo bridged NiII–NiII motifs that is preserved following a symmetric contraction of Ni–O bond lengths when oxidized to di-μ-oxo NiIV–NiIV. Incorporation of Fe ions into the structure generates di-μ-hydroxo NiII–FeIII motifs in which Ni–Fe distances are dependent on nickel oxidation state, but Fe–O bond lengths are not. This asymmetry minimizes the trigonal distortion in di-μ-hydroxo NiII–FeIII motifs and neighboring di-μ-hydroxo NiII–NiII sites in the reduced state, but exacerbates it in the oxidized state. We attribute both the Fe-induced anodic shift in nickel-based redox peaks and the improved ability to catalyze the oxygen evolution reaction to this inversion in geometric distortions. Spectroelectrochemical experiments reveal a previously unreported change in optical absorbance at ca. 1.5 V vs. RHE in Fe-containing samples. We attribute this feature to oxidation of nickel ions in di-μ-hydroxo NiII–FeIII motifs, which we propose is the process relevant to catalytic oxygen evolution.


 Please wait while we load your content...
Please wait while we load your content...