Pyro-catalytic hydrogen evolution by Ba0.7Sr0.3TiO3 nanoparticles: harvesting cold–hot alternation energy near room-temperature
Abstract
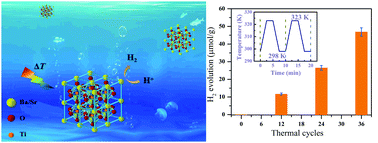
Photocatalytic hydrogen evolution, as an environmental-friendly, energy-saving and simple process, has been widely reported. However, there are some important factors that prohibit practical application of photocatalytic hydrogen production, including the low efficiency of solar energy utilization, and the lack of response in the dark. Temperature fluctuation is a very common phenomenon in our daily life, which is rarely reported for use for hydrogen production. Here we demonstrate direct hydrogen evolution through the harvesting of cold–hot 298–323 K (25–50 °C) alternation energy by using pyroelectric nanomaterial Ba0.7Sr0.3TiO3 (BST). The yield of hydrogen achieved was 46.89 μmol per gram of catalyst after 36 thermal cycles and the rate of hydrogen generation was up to 1.30 μmol g−1 per thermal cycle. The minimum of the conduction band of BST is more negative than the conduction band level of H+/H2, enabling the reduction of water to form hydrogen. This efficient and eco-friendly pyro-catalytic method provides a promising future for hydrogen evolution utilizing natural cold–hot temperature fluctuation energy.


 Please wait while we load your content...
Please wait while we load your content...