Elucidating lithium-ion and proton dynamics in anti-perovskite solid electrolytes†
Abstract
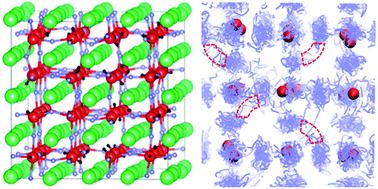
All-solid-state Li-ion batteries are currently attracting considerable research attention as they present a viable opportunity for increased energy density and safety when compared to conventional liquid electrolyte-based devices. The Li-rich anti-perovskite Li3−xOHxCl has generated recent interest as a potential solid electrolyte material, but its lithium and proton transport capabilities as a function of composition are not fully characterised. In this work, we apply a combination of ab initio molecular dynamics and 1H, 2H and 7Li solid-state NMR spectroscopy to study the mobility of lithium ions and protons in Li3−xOHxCl. Our calculations predict a strongly exothermic hydration enthalpy for Li3OCl, which explains the ease with which this material absorbs moisture and the difficulty in synthesising moisture-free samples. We show that the activation energy for Li-ion conduction increases with increasing proton content. The atomistic simulations indicate fast Li-ion diffusion but rule out the contribution of long-range proton diffusion. These findings are supported by variable-temperature solid-state NMR experiments, which indicate localised proton motion and long-range Li-ion mobility that are intimately connected. Our findings confirm that Li3−xOHxCl is a promising solid electrolyte material for all-solid-state Li-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...