Boosting the hydrogen evolution performance of ruthenium clusters through synergistic coupling with cobalt phosphide†
Abstract
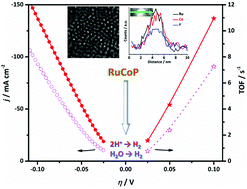
In this article, we for the first time report the synthesis and electrocatalytic properties of ruthenium cobalt phosphide hybrid clusters for the hydrogen evolution reaction (HER). Two types of catalysts are investigated: wet chemical reduction of Ru3+ on pre-formed cobalt phosphide (CoP) nanoparticles results in ruthenium–cobalt phosphide side-by-side structures (Ru/CoP); while phosphorizing chemically reduced RuCo alloy leads to the formation of hybrid ruthenium cobalt phosphide (RuCoP) clusters. Compared to pristine Ru clusters, both Ru/CoP and RuCoP show significantly improved HER performance in both acidic and alkaline solutions. In particular, the hybrid RuCoP clusters demonstrate a considerably low overpotential (η10) of 11 mV at −10 mA cm−2 and a high turnover frequency (TOF) of 10.95 s−1 at η = 100 mV in 0.5 M H2SO4. Even in 1.0 M KOH the excellent HER activity of the RuCoP clusters remains, with a very low η10 of 23 mV and exceptionally high TOF value of 7.26 s−1 at η = 100 mV. Moreover, the RuCoP catalysts can sustain galvanostatic electrolysis in both acidic and alkaline solutions at −10 mA cm−2 for 150 h with little degradation, showing better catalytic stability than the state-of-the-art commercial Pt/C catalysts. Our density functional theory (DFT) calculations indicate that the RuCoP hybrid exhibits a hydrogen adsorption energy very close to that of Pt and water and –OH adsorption energies distinct from pristine Ru, which reasonably explain the experimentally observed excellent HER activities and highlight the importance of synergistic coupling with cobalt phosphide to boost the HER performance of ruthenium.


 Please wait while we load your content...
Please wait while we load your content...