Isolated Ni single atoms in graphene nanosheets for high-performance CO2 reduction†
Abstract
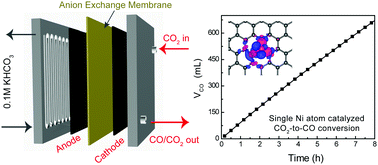
Single-atom catalysts have emerged as an exciting paradigm with intriguing properties different from their nanocrystal counterparts. Here we report Ni single atoms dispersed into graphene nanosheets, without Ni nanoparticles involved, as active sites for the electrocatalytic CO2 reduction reaction (CO2RR) to CO. While Ni metal catalyzes the hydrogen evolution reaction (HER) exclusively under CO2RR conditions, Ni single atomic sites present a high CO selectivity of 95% under an overpotential of 550 mV in water, and an excellent stability over 20 hours’ continuous electrolysis. The current density can be scaled up to more than 50 mA cm−2 with a CO evolution turnover frequency of 2.1 × 105 h−1 while maintaining 97% CO selectivity using an anion membrane electrode assembly. Different Ni sites in graphene vacancies, with or without neighboring N coordination, were identified by in situ X-ray absorption spectroscopy and density functional theory calculations. Theoretical analysis of Ni and Co sites suggests completely different reaction pathways towards the CO2RR or HER, in agreement with experimental observations.


 Please wait while we load your content...
Please wait while we load your content...