Constructing canopy-shaped molecular architectures to create local Pt surface sites with high tolerance to H2S and CO for hydrogen electrooxidation†
Abstract
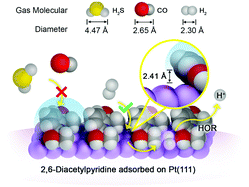
Rational design and construction of the local environment of active sites on noble metal surfaces is a promising, but challenging, approach for developing high-selectivity catalysts. This study presents an effective approach, via engineering local active sites, aiming to solve the critical problem of H2S and CO poisoning of Pt catalysts for H2 electrooxidation, the anode reaction of polymer electrolyte membrane fuel cells. A canopy-shaped molecular architecture was constructed by immobilizing an organic molecule, 2,6-diacetylpyridine (DAcPy), on Pt surface, which exhibits high H2S (1 ppm) and CO (100 ppm) tolerance. Through electrochemical, spectroscopic, and DFT studies, as well as comparative investigation of analogous structure molecules, it was revealed that DAcPy can be strongly adsorbed on Pt surface through tridentate coordination (two Pt–C and one Pt–N bonds), allowing it to compete with CO and H2S adsorption. The pyridine ring of DAcPy is in a tilted orientation, providing some protection underneath the ring for Pt atoms. Such a height-limited space is just accessible for small-sized H2, but not for relatively large H2S and CO. This study demonstrates that regulating steric hindrance to protect active sites is a promising approach for designing highly selective electrocatalysts.
- This article is part of the themed collection: 2018 Energy and Environmental Science HOT Articles


 Please wait while we load your content...
Please wait while we load your content...