Solvent-dependent transition from concerted electron–proton to proton transfer in photoinduced reactions between phenols and polypyridine Ru complexes with proton-accepting sites†
Abstract
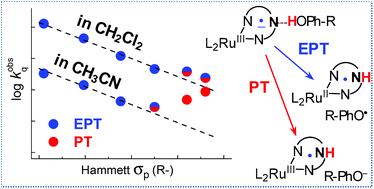
The bimolecular rate coefficients (kobsq) for quenching the metal-to-ligand charge transfer excited states of two Ru polypyridine complexes containing H-bond accepting sites by six p-substituted phenols exhibit abrupt deviations from the expected linear correlations of log kobsq with phenol's Hammett σp constant. This pattern is attributed to a transition of the quenching mechanism from a concerted electron–proton transfer (EPT) to a proton transfer (PT); the latter becomes predominant for the most acidic phenols in acetonitrile, but not in dichloromethane. This assertion is supported by a detailed thermochemical analysis, which also excludes the quenching pathways involving electron transfer from phenols with or without deprotonation of phenols to the solvent, either concerted or sequential. The transition from EPT to PT upon the σp increase is consistent/supported by the magnitudes of the measured and computed PhOH/OD kinetic isotope effects and by the observed reduction of the EPT product yields upon replacing the low σp methoxyphenol by the high σp nitrophenol. In addition to modulating the relative contribution of the EPT and PT quenching pathways, the solvent strongly affects the bimolecular rate coefficients for the EPT quenching proper. Unlike with H-atom transfer reactions, this kinetic solvent effect could not be quantitatively accounted for by the phenol-solvent H-bonding alone, which suggests a solvent effect on the H-bonding constants in the phenol-Ru complex precursor exciplexes and/or on the unimolecular EPT rate coefficients within these exciplexes.


 Please wait while we load your content...
Please wait while we load your content...
