Mixed chelating ligands used to regulate the luminescence of Ln(iii) complexes and single-ion magnet behavior in Dy-based analogues†
Abstract
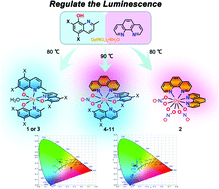
The organic ligands 5,7-dibromo-2-methyl-8-quinolinol (L1), 1,10-phenanthroline (L2), and 5,7-dichloro-2-methyl-8-quinolinol (L3) were used to react with Dy(NO3)3·6H2O under solvothermal conditions at 80 °C to obtain the complexes [Dy(L1)3(H2O)] (1), [Dy(L2)2(NO3)3] (2), and [Dy(L3)3(H2O)] (3), respectively. The reaction of L1 and L2 with lanthanide(III) nitrate salts in the presence of triethylamine as a base afforded four mononuclear complexes, namely, [Ln(L1)2(L2)(NO3)] [Ln = Dy (4), Ho (5), Er (6), and Tb (7)]. Complexes 1 and 2 emitted yellow-green and red light under excitation with light of a certain wavelength. Interestingly, 4–7 exhibited a superimposition of the luminescence of 1 and 2. To our knowledge, this is the first example of the use of different organic light-emitting ligands to adjust the fluorescence of Ln(III) complexes. Moreover, the series of complexes [Ln(L3)2(L2)(NO3)] [Ln = Dy (8), Ho (9), Er (10), and Tb (11)] were also obtained under the same conditions by replacing L1 with L3. In the way that was expected, 8–11 exhibited a superimposition of the luminescence of 2 and 3. Density functional theory (DFT) calculations of electron cloud density showed that the electron cloud densities of complexes 4 and 8 are mainly concentrated in the quinoline rings. Furthermore, analysis of the molecular ion peaks of complexes 4–11 obtained by electrospray mass spectrometry (ESI-MS) showed that only the 1,10-phenanthroline ligand was discovered to dissociate in the solution state. Magnetic measurements of the Dy-containing complexes revealed features of field-induced single-ion magnet behavior.


 Please wait while we load your content...
Please wait while we load your content...