Solid-state 45Sc NMR studies of Cp*2Sc–X and Cp*2ScX(THF)†
Abstract
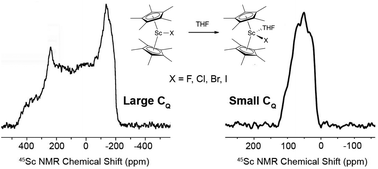
Cp*2Sc–X, where X is a halide, were synthesized and studied by solid-state 45Sc NMR to determine how the Sc–X bond affects quadrupolar NMR parameters. The experimental quadrupolar coupling constants (CQ) show that the fluoride has the largest coupling constant and that the iodide has the smallest coupling constant. DFT analysis of this data indicates that the CQ of these compounds is related to core scandium and halide orbitals, which is related to polarizability of the halide and the Sc–X distance. Cp*2ScX(THF) were also investigated by solid-state 45Sc NMR spectroscopy, and have much smaller CQ values than the base-free halides. This is related to the change in structure of the THF adduct and occupation of orbitals of π-symmetry that reduce CQ.
- This article is part of the themed collection: In celebration of Richard Andersen’s 75th birthday


 Please wait while we load your content...
Please wait while we load your content...