Synthesis and structural characterisation of unprecedented primary N-nitrosamines coordinated to iridium(iv)†
Abstract
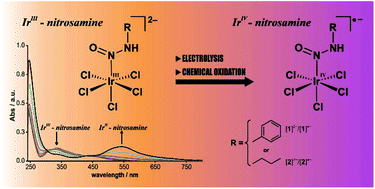
The redox chemistry of the N-nitrosamine complexes [IrCl5(RN(H)N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)]2– (R = benzyl or n-butyl) was studied in detail. One-electron oxidations at around 200 mV vs. ferrocene/ferrocenium were reversible in cyclic voltammograms. UV-vis spectroelectrochemistry reveals spectra characteristic of IrIV species but also partial decomposition of the oxidised species [IrIVCl5(RN(H)N
O)]2– (R = benzyl or n-butyl) was studied in detail. One-electron oxidations at around 200 mV vs. ferrocene/ferrocenium were reversible in cyclic voltammograms. UV-vis spectroelectrochemistry reveals spectra characteristic of IrIV species but also partial decomposition of the oxidised species [IrIVCl5(RN(H)N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)]˙− on this timescale (minutes). Detailed studies on chemically oxidised solutions of the parent IrIII complexes gave evidence for paramagnetic IrIV from NMR spectra. Final products of the decomposition were the corresponding alcohols and presumably [IrIIICl5(L)]2– (L = N2, solvent, amine) complexes. Similar decomposition reactions of acidic DMSO solutions of [IrCl5(RN(H)N
O)]˙− on this timescale (minutes). Detailed studies on chemically oxidised solutions of the parent IrIII complexes gave evidence for paramagnetic IrIV from NMR spectra. Final products of the decomposition were the corresponding alcohols and presumably [IrIIICl5(L)]2– (L = N2, solvent, amine) complexes. Similar decomposition reactions of acidic DMSO solutions of [IrCl5(RN(H)N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)]2–revealed that this combination produces the so-called “activated” DMSO (Me2S+–O− or Me2S+–OE, with “E” being an electrophile) which oxidises the parent IrIII complexes. Finally, with the very reactive purple IrIV compound (PPh4)[IrCl5(BnN(H)N
O)]2–revealed that this combination produces the so-called “activated” DMSO (Me2S+–O− or Me2S+–OE, with “E” being an electrophile) which oxidises the parent IrIII complexes. Finally, with the very reactive purple IrIV compound (PPh4)[IrCl5(BnN(H)N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)], the first primary N-nitrosamine coordinated to [IrIVCl5]− was isolated and characterised by UV-vis absorption, FTIR, NMR spectroscopy, ultra-high resolution electrospray mass spectrometry (UHR-ESI-MS) and iridium L3 X-ray absorption near-edge spectroscopy (XANES).
O)], the first primary N-nitrosamine coordinated to [IrIVCl5]− was isolated and characterised by UV-vis absorption, FTIR, NMR spectroscopy, ultra-high resolution electrospray mass spectrometry (UHR-ESI-MS) and iridium L3 X-ray absorption near-edge spectroscopy (XANES).


 Please wait while we load your content...
Please wait while we load your content...