Compositional and structural insights into the nature of the H-cluster precursor on HydF†
Abstract
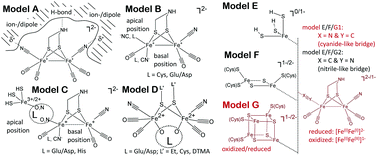
Assembly of an active [FeFe]-hydrogenase requires dedicated maturation enzymes that generate the active-site H-cluster: the radical SAM enzymes HydE and HydG synthesize the unusual non-protein ligands – carbon monoxide, cyanide, and dithiomethylamine – while the GTPase HydF serves as a scaffold for assembly of the 2Fe subcluster containing these ligands. In the current study, enzymatically cluster-loaded HydF ([2Fe]F) is produced by co-expression with HydE and HydG in an Escherichia coli host followed by isolation and examination by FTIR and EPR spectroscopy. FTIR reveals the presence of well-defined terminal CO and CN− ligands; however, unlike in the [FeFe]-hydrogenase, no bridging CO is observed. Exposure of this loaded HydF to exogenous CO or H2 produces no significant changes to the FTIR spectrum, indicating that, unlike in the [FeFe]-hydrogenase, the 2Fe cluster in loaded HydF is coordinatively saturated and relatively unreactive. EPR spectroscopy reveals the presence of both [4Fe–4S] and [2Fe–2S] clusters on this loaded HydF, but provides no direct evidence for these being linked to the [2Fe]F. Using the chemical reactivity and FTIR data, a large collection of computational models were evaluated. Their scaled quantum chemical vibrational spectra allowed us to score various [2Fe]F structures in terms of their ability to reproduce the diatomic stretching frequencies observed in the FTIR experimental spectra. Collectively, the results provide new insights that support the presence of a diamagnetic, but spin-polarized FeI–FeI oxidation state for the [2Fe]F precursor cluster that is coordinated by 4 CO and 2 CN− ligands, and bridged to an adjacent iron–sulfur cluster through one of the CN− ligands.


 Please wait while we load your content...
Please wait while we load your content...