Polycarboxylate-directed semi-rigid pyridyl-amide-based various NiII complexes: electrochemical properties and enhancements of photocatalytic activities by calcination†
Abstract
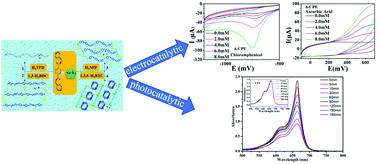
In this work, four new NiII coordination polymers (CPs) based on a bi-methylene-bridged semi-rigid bis-pyridyl-bis-amide ligand and various polycarboxylates, namely, [Ni(4-bmbpd)(H2O)4]·(TPD) (1), [Ni2(4-bmbpd)2(1,3-BDC)2(H2O)4]·5H2O (2), [Ni2(4-bmbpd)2(MIP)2(H2O)2]·3H2O (3) and [Ni3(4-bmbpd)3(BTC)2(H2O)4]·4H2O (4) [4-bmbpd = N,N′-bis(4-methylenepyridin-4-yl)-1,4-benzenedicarboxamide, H2TPD = 2,5-thiophenedicarboxylic acid, 1,3-H2BDC = 1,3-benzenedicarboxylic acid, H2MIP = 5-methylisophthalic acid and 1,3,5-H3BTC = 1,3,5-benzenetricarboxylic acid] have been hydrothermally synthesized and structurally characterized by single-crystal X-ray diffraction analyses, thermal gravimetric (TG) analyses, IR spectroscopy and powder X-ray diffraction (PXRD). In complex 1, the μ2-bridging 4-bmbpd ligands connect adjacent NiII ions leading to a 1D [Ni-4-bmbpd]n wave-like chain, while the TPD anions act as discrete counter ions. Complexes 2 and 3 are similar 2D 4-connected grid networks derived from [Ni-4-bmbpd]n chains and [Ni-L]n (L = BDC or MIP) chains. 4 shows a 3D (2,3,4,4)-connected framework, which contains [Ni-1,3,5-BTC]n 1D double chains, [Ni-(4-bmbpd)]n linear chains and [Ni2(4-bmbpd)2] rings. The 1D chains for 1 and the 2D networks for 2 and 3 are finally extended into 3D supramolecular architectures by hydrogen bonding interactions. The role of polycarboxylates in the assembly and structures of the target compounds was discussed. Moreover, photoluminescence properties, electrochemical properties of complexes 1–4 and the photocatalytic activities of calcination products of 1 and 4 (Ni@NC1 and Ni@NC4) were investigated. More importantly, the photocatalytic performances of Ni@NC1 and Ni@NC4 are enhanced compared with the title complexes 1 and 4.


 Please wait while we load your content...
Please wait while we load your content...