Synthesis, structure, and polymorphic transitions of praseodymium(iii) and neodymium(iii) borohydride, Pr(BH4)3 and Nd(BH4)3†
Abstract
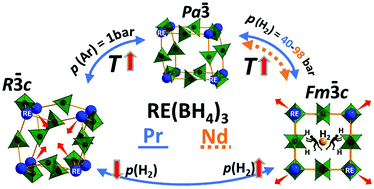
In this work, praseodymium(III) borohydride, Pr(BH4)3, and an isotopically enriched analogue, Pr(11BD4)3, are prepared by a new route via a solvate complex, Pr(11BD4)3S(CH3)2. Nd(BH4)3 was synthesized using the same method and the structures, polymorphic transformations, and thermal stabilities of these compounds are investigated in detail. α-Pr(BH4)3 and α-Nd(BH4)3 are isostructural with cubic unit cells (Pa![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) stable at room temperature (RT) and a unit cell volume per formula unit (V/Z) of 180.1 and 175.8 Å3, respectively. Heating α-Pr(BH4)3 to T ∼ 190 °C, p(Ar) = 1 bar, introduces a transition to a rhombohedral polymorph, r-Pr(BH4)3 (R
) stable at room temperature (RT) and a unit cell volume per formula unit (V/Z) of 180.1 and 175.8 Å3, respectively. Heating α-Pr(BH4)3 to T ∼ 190 °C, p(Ar) = 1 bar, introduces a transition to a rhombohedral polymorph, r-Pr(BH4)3 (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c) with a smaller unit cell volume and a denser structure, V/Z = 156.06 Å3. A similar transition was not observed for Nd(BH4)3. However, heat treatment of α-Pr(BH4)3, at T ∼ 190 °C, p(H2) = 40 bar and α-Nd(BH4)3, at T ∼ 270 °C, p(H2) = 98 bar facilitates reversible formation of another three cubic polymorph, denoted as β, β′ and β′′-RE(BH4)3 (Fm
c) with a smaller unit cell volume and a denser structure, V/Z = 156.06 Å3. A similar transition was not observed for Nd(BH4)3. However, heat treatment of α-Pr(BH4)3, at T ∼ 190 °C, p(H2) = 40 bar and α-Nd(BH4)3, at T ∼ 270 °C, p(H2) = 98 bar facilitates reversible formation of another three cubic polymorph, denoted as β, β′ and β′′-RE(BH4)3 (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c). Moreover, the transition β- to β′- to β′′- is considered a rare example of stepwise negative thermal expansion. For Pr(BH4)3, ∼2/3 of the sample takes this route of transformation whereas in argon only ∼5 wt%, and the remaining transforms directly from α- to r-Pr(BH4)3. The β-polymorphs are porous with V/Z = 172.4 and 172.7 Å3 for β′′-RE(BH4)3, RE = Pr or Nd, respectively, and are stabilized by the elevated hydrogen pressures. The polymorphic transitions occur due to rotation of RE(BH4)6 octahedra without breaking or forming chemical bonds. Structural DFT optimization reveals the decreasing stability of α-Pr(BH4)3 > β-Pr(BH4)3 > r-Pr(BH4)3.
c). Moreover, the transition β- to β′- to β′′- is considered a rare example of stepwise negative thermal expansion. For Pr(BH4)3, ∼2/3 of the sample takes this route of transformation whereas in argon only ∼5 wt%, and the remaining transforms directly from α- to r-Pr(BH4)3. The β-polymorphs are porous with V/Z = 172.4 and 172.7 Å3 for β′′-RE(BH4)3, RE = Pr or Nd, respectively, and are stabilized by the elevated hydrogen pressures. The polymorphic transitions occur due to rotation of RE(BH4)6 octahedra without breaking or forming chemical bonds. Structural DFT optimization reveals the decreasing stability of α-Pr(BH4)3 > β-Pr(BH4)3 > r-Pr(BH4)3.


 Please wait while we load your content...
Please wait while we load your content...