Diastereoselective synthesis, structure and reactivity studies of ferrocenyloxazoline gold(i) and gold(ii) complexes†
Abstract
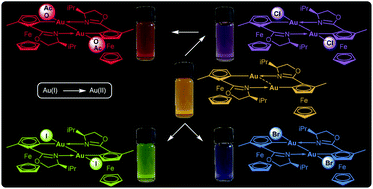
In the last few decades, gold complexes have demonstrated huge potential for soft Lewis acid catalysis. Despite the intensive research on Au complexes and planar chiral metallacycles, enantiopure ferrocenylgold complexes have – surprisingly – not been reported until the studies presented in this article. Herein, we report the asymmetric synthesis of planar chiral ferrocenyl Au(I) complexes. These dinuclear species form helically chiral ten-membered (NCCCAu)2 rings stabilized by aurophilic interactions. In supramolecular solid state structures, linear, zigzag or helical Au(I) wires with regular Au⋯Au separations were observed. The dissolved dinuclear entities could be oxidized by Au(I) to unique ferrocenyl Au(II) complexes featuring short Au(II)–Au(II) bonds, while the ferrocene core remained intact. However, our initial studies revealed the issue of configurational lability of the ferrocenyl Au(II) complexes in terms of the element of planar chirality in the presence of the gold source, (Me2S)AuCl. This was successfully addressed by a systematic study implementing permanent σ-donor ortho-protecting groups such as methyl and trimethylsilyl, which impede an epimerization event. Oxidation of the dinuclear Au(I) complexes was also accomplished by oxidative addition reactions with halogenated solvents, preferably CHCl3. Additional reactivity studies revealed that dinuclear Au(II) dihalide complexes are also formed with reactive alkylhalides such as iodomethane, benzylbromide and benzyliodide. Interestingly, the whole spectral range of colors (violet, blue, green, yellow, and red) is covered by the title complexes depending on the Au oxidation state and the anionic ligands in the Au(II) complexes. This appears to be quite unusual for ferrocenes, which typically adopt orange to red colors in a non-oxidized state.


 Please wait while we load your content...
Please wait while we load your content...