Bromination and iodination of diphosphane dichalcogenides†
Abstract
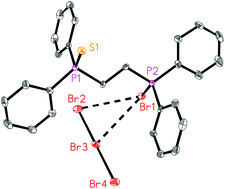
The diphosphane dichalcogenides dppmSe2, dppeSe2, diprpmSe2, diprpeSe2, dppmS2 and dppeS2, [dppm = bis(diphenylphosphano)methane, dppe = bis(diphenylphosphano)ethane, diprpm = bis(diisopropylphosphano)methane, diprpe = bis(diisopropylphosphano)ethane] have been treated with elemental bromine or iodine and the products 1–15 characterized by single crystal X-ray diffraction. DppeSe2 with bromine or iodine gives the simple (and previously known) adducts dppe(SeBr2)21 or dppe(SeI2)22 respectively; diprpeSe2 reacts similarly to give diprpe(SeBr2)23 (at 0 °C) or diprpe(SeI2)24. The bromine derivatives display a T-shaped geometry at selenium, whereas the iodine derivatives involve linear Se–I–I groups. With dppmSe2 and bromine, a product was formed at −50 °C, but could not be isolated; at room temperature the reaction leads to a mixture of products, from which only [dppmBr2]2+ [Se2Br10]2–5 (the anion involving a dibromine molecule coordinating to a Se centre) could be isolated. Reaction of dppeSe2 with excess bromine at room temperature leads to [dppeBr2]2+·½[SeBr6]2–·[SeBr2·Br2·Br3]−6. The reaction of dppmSe2 with iodine gives the 1 : 2 adduct [dppmSe2I]+ [I3]−7, the cation of which contains a novel five-membered CP2Se2 heterocycle. In a 1 : 1 ratio the same reaction gives another 1 : 2 adduct, [I2Se(dppm)Se–I–Se(dppm)SeI2]+ [I3]−8. DiprpmSe2 with iodine gives [diprpmSe2I]+ [I3]−9, analogous to 7, and with bromine at 0 °C the corresponding product [diprpmSe2Br]+ [Br3]−10. At room temperature, diprpeSe2 reacts with bromine to give a mixture of [diprpe(SeBr2)Br]+ Br−11 and [diprpeBr2]2+ [SeBr4]2–12, whereas diprpmSe2 forms [diprpmBr2]2+ [SeBr4]2–13, analogous to 12. The disulfides dppmS2 and dppeS2 react with bromine to give [dppmSBr]+ [Br3]−14 and [dppeSBr]+ [Br3]−15.

 Please wait while we load your content...
Please wait while we load your content...