Platinum(iv) azido complexes undergo copper-free click reactions with alkynes†
Abstract
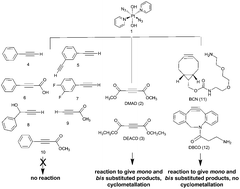
We report our investigations into the first examples of copper-free 1,3-dipolar cycloaddition (click) reactions of electrophiles with a PtIV azido complex. The Pt-IV azido complex trans, trans, trans-[PtIV(py)2(N3)2(OH)2] (1) was reactive towards dimethyl acetylenedicarboxylate (DMAD) (2), diethyl acetylenedicarboxylate DEACD (3), N-[(1R,8S,9s)-bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl]-1,8-diamino-3,6-dioxaoctane (BCN) (11) and dibenzocyclooctyne-amine (DBCO) (12) resulting in formation of the corresponding mono (a) and bis-substituted (b) complexes. Complexes of 2 undergo further reactions between the Pt centre and the carbonyl group to form 2a′ and 2b′. This is not seen for the products of the corresponding PtII azido complex trans-[Pt(py)2(N3)2] with acetylene 2. Novel complexes 2a′, 2b′, 11a and 11b have been characterised by multinuclear NMR, IR and UV-vis spectroscopy and ESI-MS. These reactions represent new synthetic routes to novel Pt(IV) complexes.
- This article is part of the themed collection: New Talent: Europe


 Please wait while we load your content...
Please wait while we load your content...