Synthesis and characterization of a novel aminopolycarboxylate complexant for efficient trivalent f-element differentiation: N-butyl-2-acetamide-diethylenetriamine-N,N′,N′′,N′′-tetraacetic acid†
Abstract
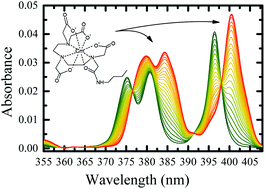
The novel metal ion complexant N-butyl-2-acetamide-diethylenetriamine-N,N′,N′′,N′′-tetraacetic acid (DTTA-BuA) uses an amide functionalization to increase the total ligand acidity and attain efficient 4f/5f differentiation in low pH conditions. The amide, when located on the diethylenetriamine platform containing four acetate pendant arms maintains the octadentate coordination sphere for all investigated trivalent f-elements. This compact coordination environment inhibits the protonation of LnL− complexes, as indicated by lower K111 constants relative to the corresponding protonation site of the free ligand. For actinide ions, the enhanced stability of AnL− lowers the K111 for americium and curium beyond the aptitude of potentiometric detection. Density functional theory computations indicate the difference in the back-donation ability of Am3+ and Eu3+ f-orbitals is mainly responsible for stronger proton affinity of EuL− compared to AmL−. The measured stability constants for the formation of AmL− and CmL− complexes are consistently higher, relative to ML− complexes with lanthanides of similar charge density. When compared with the conventional aminopolycarboxylate diethylenetriamine pentaacetic acid (DTPA), the modified DTTA-BuA complexant features higher ligand acidity and the important An3+/Ln3+ differentiation when deployed on a liquid–liquid distribution platform.


 Please wait while we load your content...
Please wait while we load your content...