Neuroprotective alpha-cleavage of the human prion protein significantly impacts Cu(ii) coordination at its His111 site†
Abstract
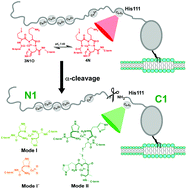
The cellular prion protein (PrPC) is a copper binding protein that undergoes post-translational modifications, such as endoproteolytic alpha cleavage, which occurs in the vicinity of the His111 Cu binding site. Alpha cleavage processing of PrPC is considered to be neuroprotective since the cleavage site is located in a region that is key to the conversion of PrPC into the infectious scrapie isoform (PrPSc), yielding a membrane bound C1 fragment of PrPC that still contains His111. In this work, we use hPrP(111–115) fragment as a model peptide to evaluate the impact of alpha cleavage processing of PrPC in its ability to coordinate Cu(II) ions at His111. By using different spectroscopic techniques such as electronic absorption, circular dichroism, nuclear magnetic resonance, and electron paramagnetic resonance, this study demonstrates that Cu(II) binding to the cleaved His111 site is highly dependent on Cu and proton concentrations. The imidazole group of His111 and its free NH2 terminus emerge as the main anchoring sites for Cu(II) coordination, yielding very different complexes from those characterized for the intact His111 site in the full protein. Different Cu(II) coordination modes that could form with the alpha cleaved PrPC under physiological conditions are identified and characterized. Overall, this study contributes to understand how alpha cleavage processing of PrPC impacts its Cu(II) binding properties at His111. While the functional implications of Cu binding to the cleaved PrPC remain to be discovered, proteolytic processing of PrPC and its Cu binding features appear to be molecular events that might be strongly linked to its cellular function.
- This article is part of the themed collections: Celebrating recent chemical science in Mexico and Frontiers in Spectroscopic Techniques in Inorganic Chemistry


 Please wait while we load your content...
Please wait while we load your content...