Chiral hybrid materials based on pyrrolidine building units to perform asymmetric Michael additions with high stereocontrol†
Abstract
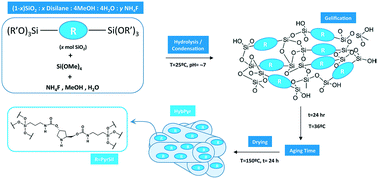
A new chiral mesoporous hybrid material was synthesized based on pyrrolidine units included in a siliceous framework, HybPyr, and integrated into the organic–inorganic structure, from a specific bis-silylated precursor. A fluoride sol–gel methodology under soft synthesis conditions and in the absence of sophisticated structural directing agents allowed the generation of a mesoporous architecture with a homogeneous distribution of active chiral moieties along the network. The hybrid material was studied by means of different characterization techniques (TGA, NMR and IR spectroscopy, chemical and elemental analyses, TEM, and textural measurements), verifying the stability and integrity of the asymmetric active sites in the solid. The hybrid material, HybPyr, is an excellent asymmetric heterogeneous and recyclable catalyst for enantioselective Michael addition of linear aldehydes to β-nitrostyrene derivatives with high stereocontrol of the reaction products.


 Please wait while we load your content...
Please wait while we load your content...