Manipulating the mesostructure of silicoaluminophosphate SAPO-11 via tumbling-assisted, oriented assembly crystallization: a pathway to enhance selectivity in hydroisomerization†
Abstract
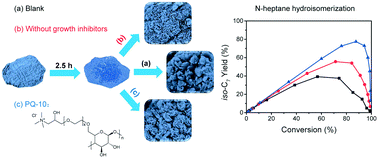
Controlling the architecture of crystalline materials via a non-classical crystallization pathway provides a versatile route to optimise their properties. It is shown how a designed hierarchical architecture of silicoaluminophosphate SAPO-11 can be obtained by controlling the crystallization process, and how this affects hydroisomerization of n-heptane. Oriented attachment is identified as the main crystallization pathway when pre-fabricated nanocrystallites are used as precursors. Tumbling crystallization facilitates the in-plane alignment of nanocrystallites to afford a house-of-cards architecture, consisting of ca. 300 nm nanosheets along the same crystallographic b axis, while static crystallization only affords randomly oriented agglomerates made up of fused nanocrystallites. Using a growth modifier, cellulose 2-(2-hydroxy-3-(trimethylammonium)propoxy) ethyl ether chloride (polyquaternium-10 or PQ-10), reduces the lateral size of these platelets down to ca. 20 nm, while simultaneously increasing the auxiliary porosity and surface area, as well as the mass transfer properties of the hierarchically structured material. The preparation factors that influence the morphology of the final crystalline products can be associated with changes in interparticle interactions. A suite of characterization techniques, such as XRD, N2 physisorption, Hg porosimetry, SEM, TEM, 28Al, 31P and 29Si MAS NMR, NH3-TPD and Py-IR were employed to elucidate the structure and acidity. Catalytic hydroisomerization tests for n-heptane demonstrate that the selectivity to isomerized components is governed by diffusion properties that rely on a hierarchical architecture. Hydroisomerization is the main reaction pathway only when operated within the diffusion controlled regime. Our findings thus provide more insight into how the hierarchical architecture of a catalyst influences the reaction product distribution.


 Please wait while we load your content...
Please wait while we load your content...