Spectroscopic characterization of a highly selective NiCu3/C hydrodeoxygenation catalyst†
Abstract
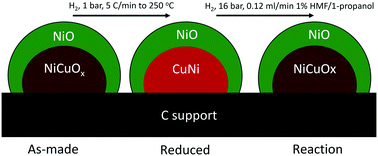
The design of selective and active heterogeneous catalysts for the hydrodeoxygenation (HDO) of biomass-derived platform molecules requires understanding of the catalyst structure under reaction conditions. In this work, we design, build and qualify an operando X-ray absorption (XAS) cell to characterize a highly selective NiCu3 catalyst under HDO conditions. We complement XAS measurements with X-ray photoelectron spectroscopy (XPS) studies and theoretical calculations to show that in bimetallic NiCu catalysts, Ni and Cu are segregated. The as-made catalysts are mixed oxides, with a NiO shell over a NiCuOx core. The core becomes a partially reduced metal alloy after treatment with hydrogen at temperatures below 473 K, while the shell becomes fully reduced at higher treatment temperatures. The liquid-phase reaction results in gradual reoxidation of the catalyst to a core–shell mixed oxide, in which the Cu in the core is predominately Cu(I) and the Ni shell is predominately Ni(II).


 Please wait while we load your content...
Please wait while we load your content...