CO oxidation over MOx (M = Mn, Fe, Co, Ni, Cu) supported on SmMn2O5 composite catalysts†
Abstract
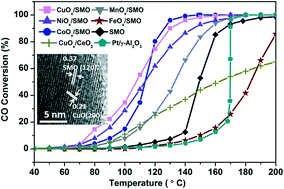
The ability to oxidize CO at relatively low temperature is barely satisfactory for pure SmMn2O5 (SMO) catalysts. While loading precious metals like platinum nanoclusters on mullite SMO is an effective approach to enhance its catalytic performance, the high cost of noble metals has limited its large scale application. In this work, we systematically study an alternative approach to composite catalyst formation by loading non-precious metal oxides MOx (M = Mn, Fe, Co, Ni, Cu) onto SMO mullite for CO oxidation. Inductively coupled plasma-optical emission spectroscopy (ICP-OES) and high-resolution transmission electron microscopy (HRTEM) analyses show that composite catalysts are successfully formed by loading MOx nanoparticles at a content of ∼0.75 wt% through the deposition precipitation (DP) method. Except for FeOx, MOx loaded samples in general exhibit much higher reaction rates of CO oxidation than bare SMO especially at the temperature range below 100 °C. The promoted activity for MOx/SMO composites can be attributed to the improved reducibility of adsorbed oxygen (Oads) and the increased oxidation state of Mn cations at the interface. Such behavior accelerates the decomposition of carbonates, as indicated by the H2 temperature-programmed reduction (TPR), X-ray photoelectron spectroscopy (XPS) and diffuse reflectance infrared Fourier transform (DRIFT) spectroscopy results. We demonstrate that the best catalytic performance of the CuOx/SMO composite also stems from the additional CO adsorption on the CuOx sites. The work presented herein provides a method to enhance the CO oxidation performance of SMO and helps in understanding the oxidation reaction mechanism involving mullites with loaded transition metal oxides (TMOs).


 Please wait while we load your content...
Please wait while we load your content...