On the nature of active phases and sites in CO and CO2 hydrogenation catalysts
Abstract
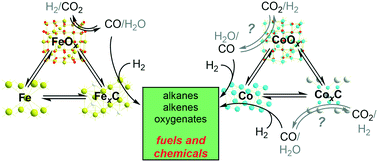
The production of hydrocarbons or oxygenates via the hydrogenation–polymerisation of either CO (Fischer–Tropsch) or CO2 as carbon sources has significant similarities from a catalytic perspective. However, strategically, the two processes differ. Fischer–Tropsch technologies, which have been commercially implemented for almost a century, rely mostly on fossil resources such as coal or natural gas after previous gasification to obtain syngas. Conversely, the direct transformation of waste CO2 using renewable hydrogen is in demand to partially compensate for its enormous emissions and to alleviate their impact on the global climate. The development of catalysts for Fischer–Tropsch processes is mature and has resulted in valuable knowledge; it also represents a valid starting point for the use of CO2 as a feedstock. In general terms, catalysts require active phases to promote (i) CO–CO2 interconversion via the water–gas shift (WGS) and its reverse (rWGS) reaction; (ii) CO activation via dissociation; (iii) hydrogenation; (iv) polymerisation leading to hydrocarbon chain growth; and (v) non-dissociative CO insertion for the formation of oxygenated moieties. Characterisation techniques have traditionally pointed to iron oxides as active phases for rWGS, to metallic cobalt or iron carbides as active phases for hydrogenation–polymerisation into hydrocarbons, and to copper, mixed cobalt–copper and rhodium as selective phases for oxygenate formation. However, the nature of these active phases and sites is still debated. Novel, improved in situ and operando investigations are challenging some previously accepted concepts by allowing the observation of metastable phases, most likely in the form of few-layer surface domains, which may play key roles. For example, the direct implication of cobalt carbides either as precursors of highly active Co(hcp) or as genuinely active phases has been proposed. Thus, the simplistic view of unique catalytically active sites is progressively being replaced with a more dynamic scenario wherein surface interconversion appears to be a common phenomenon. Herein, a perspective on the most enlightening of the recent discoveries regarding the nature of active COx hydrogenation phases and sites, providing valuable knowledge for future catalyst design, is presented.


 Please wait while we load your content...
Please wait while we load your content...