Fe–N-functionalized carbon electrocatalyst derived from a zeolitic imidazolate framework for oxygen reduction: Fe and NH3 treatment effects†
Abstract
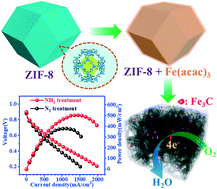
Non-precious iron–nitrogen-functionalized carbon (Fe–N–C) electrocatalysts for the oxygen reduction reaction (ORR) are prepared from ZIF-8 and iron acetylacetonate. The amount of Fe in the precursor mixture is varied to scrutinize the effect of Fe on the structural and catalytic properties of the resulting Fe–N–C. The electrocatalytic activity of the optimized sample in acidic electrolyte is comparable to that of commercial Tanaka Pt/C electrocatalyst. The catalysts were also assembled in a membrane electrode assembly as a cathode electrode for a single cell test, which shows remarkable fuel cell performance compared to other related studies of non-precious metal-based catalysts for the ORR. Such a high ORR catalytic performance can be ascribed to the synergistic effect of the Fe–N–C species, high-density pyridinic N species, N-doped carbon-encapsulated iron carbide (NC-Fe3C) nanoparticles, and high electrical conductivity as well as the porous framework structure of the as-prepared ZIF-based Fe–N–C catalysts. In addition, different properties of catalysts prepared in a nitrogen or ammonia (NH3) atmosphere are also investigated. Interestingly, it is found that NH3 treatment helps not only to improve carbon porosity and nitrogen content but also to increase carbon graphitization and induce NC-Fe3C formation, leading to enhanced ORR activity and durability of thus-prepared catalysts.


 Please wait while we load your content...
Please wait while we load your content...