Elucidating the mechanism of the UiO-66-catalyzed sulfide oxidation: activity and selectivity enhancements through changes in the node coordination environment and solvent†
Abstract
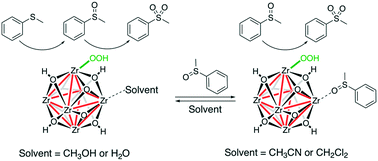
Benzoic acid modulators that “cap” the Zr6-oxo-hydroxo cluster nodes in UiO-66 metal–organic frameworks can be removed to increase the number of “open” sites (i.e., those that are terminated with [μ1-OH + μ1-OH2]) up to 5 per node, enabling the “decapped” materials to exhibit enhanced catalytic activity in the oxidation of methyl phenyl sulfide. Computational modeling reveals that the labile Zr-μ1-OH groups on these open sites are likely converted into Zr-μ1-OOH species that are active in oxidizing the sulfide as well as its sulfoxide product. In solvents such as CH3CN and CH2Cl2, the sulfoxide product can additionally replace the aquo ligands of the Zr-μ1-OH2 moieties to increase the concentration of the sulfoxide adjacent to the active Zr-μ1-OOH species, resulting in overoxidation to the sulfone. However, the use of CH3OH, a solvent that can compete with the sulfoxide and suppress this binding mode, can retard the overoxidation and lead to higher selectivities for the sulfoxide product.


 Please wait while we load your content...
Please wait while we load your content...