Optimization of N2O decomposition activity of CuO–CeO2 mixed oxides by means of synthesis procedure and alkali (Cs) promotion
Abstract
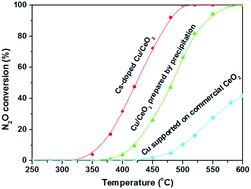
The fine-tuning of local surface chemistry of CuO–CeO2 mixed oxides by means of synthesis procedure and alkali promotion towards the rational design of highly active catalysts is investigated. In particular, the impact of alkali (Cs) promotion on the N2O decomposition activity (deN2O process) of CuO–ceria mixed oxides, pre-optimized through the preparation procedure (co-precipitation among different synthesis routes), is explored. It was found that the co-optimization of the synthesis procedure (co-precipitation) and alkali loading (1.0 at Cs per nm2) can boost the decomposition of N2O and resistance to O2 inhibition, offering a half-conversion temperature (T50) ca. 200 °C lower, when compared to a reference CuO–CeO2 sample. A complementary characterization study involving surface area determination (BET method), pore size distribution (BJH method), X-ray diffraction (XRD), H2 temperature-programmed reduction (H2-TPR) and X-ray photoelectron spectroscopy (XPS) was undertaken to gain insight into structure–property relationships. The results revealed that the superiority of Cs-doped samples can be mainly attributed to the electronic effect of the alkali promoter towards the stabilization of partially reduced Cu+/Ce3+ pairs, which play a key role in the deN2O process following a redox-type mechanism.


 Please wait while we load your content...
Please wait while we load your content...