Mechanistic insights into catalytic CO2 hydrogenation using Mn(i)-complexes with pendant oxygen ligands†
Abstract
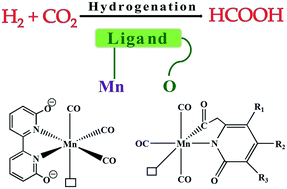
With the aid of density functional theory calculations, we have elucidated the mechanism of the catalytic hydrogenation of CO2 to formate using the recently reported dihydroxybipyridine-based Mn(I)-complex. Plausible catalytic pathways are unravelled thoroughly and the computational results account for the experimental findings. The critical roles of an external base additive and pendant hydroxyl groups, as active participants in the hydrogenation process, are examined in detail. The overall mechanism includes two main steps; heterolytic dihydrogen cleavage and hydride transfer to CO2. The initial hydrogen splitting step is assisted by the external base. Pendant oxygen atoms take part neither in the hydrogen cleavage nor directly during the subsequent hydride transfer. Nevertheless, their presence is crucial for the hydrogenation reactivity of the catalyst as they have an indirect effect in determining the feasibility of the final hydride transfer step. Furthermore, guided by detailed mechanistic understanding, we have proposed a series of Mn(I)-complexes based on an acylmethylpyridinol ligand for catalysing the hydrogenation of CO2 to formic acid.


 Please wait while we load your content...
Please wait while we load your content...