Photocatalytic oxidation of arylalcohols to aromatic aldehydes promoted by hydroxyl radicals over a CoP/CdS photocatalyst in water with hydrogen evolution†
Abstract
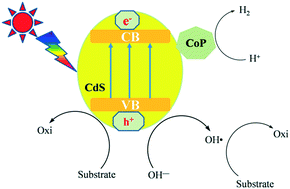
Cobalt phosphide (CoP) combined with CdS was employed as a photocatalyst to oxidize arylalcohols into aromatic aldehydes or ketones in water. This was accompanied by the reduction of water, and the quantitative yield of hydrogen evolution was much higher than the chemical equivalent of hydrogen gas. Electron spin resonance spectroscopy and quenching experiments demonstrated that hydroxyl free radicals, originating from water splitting, promoted the oxidation of arylalcohols, while the holes in the valence band of the photocatalyst were reduced by the −OH and organic substrates. The overall reaction generates high-value-added organics. This photocatalytic reaction is atom-economical, in accordance with the concept of sustainable development.


 Please wait while we load your content...
Please wait while we load your content...