Catalytic combustion of diesel soot over Fe and Ag-doped manganese oxides: role of heteroatoms in the catalytic performances†
Abstract
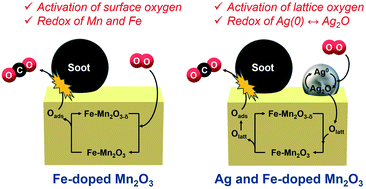
Fe-Doped manganese oxide catalysts were prepared by the citrate method and their catalytic activities in soot combustion were examined in air under tight contact mode. Among the catalysts tested, 12 mol% of Fe-doped α-Mn2O3 exhibited the highest activity, giving a soot combustion temperature (T50) of 328 °C, which is comparable to that previously reported for rare-earth-based catalysts, while pure α-Mn2O3 gave a T50 value of 339 °C. Comprehensive analyses by means of X-ray diffraction (XRD), specific surface area measurement, oxygen release rate measurement, X-ray photoelectron spectroscopy (XPS) and X-ray absorption fine structure (XAFS) measurements revealed that the improved activity was due to i) an increased specific surface area and ii) an improved oxygen release rate involving reactive surface-adsorbed oxygen species. In situ XAFS measurements suggested that the interaction between Mn and Fe facilitated the activation of the Mn–O–Fe bond and increased the proportion of reactive oxygen species, thereby improving the redox properties of the catalysts. Furthermore, simultaneous doping of Ag and Fe into Mn2O3 was examined to further enhance the catalytic activity, which gave a drastically improved catalytic activity with T50 = 290 °C. Based on temperature programmed reduction (TPR) and in situ XAFS measurements, the direct oxidation mechanism of soot by the activated lattice oxygen species via the redox of Ag0/Ag2O species was proposed.


 Please wait while we load your content...
Please wait while we load your content...