Low-temperature crystallization of solution-derived metal oxide thin films assisted by chemical processes†
Abstract
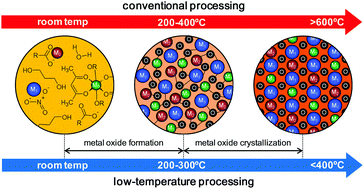
Over the last few years the efforts devoted to the research on low-temperature processing of metal oxide thin films have increased notably. This has enabled the direct integration of metal oxide layers (amorphous semiconductors) on low-melting-point polymeric substrates for flexible electronic systems, which adds to the economic and environmental benefits of the use of these processes with reduced energy consumption. More challenging is the preparation of crystalline complex oxide films at temperatures compatible with their direct integration in flexible devices. However, the usually high crystallization temperatures (>600 °C) impede the development of devices that take full advantage of the large variety of oxide functionalities available. This tutorial review analyzes a number of strategies based on wet chemical methods for inducing the crystallization of metal oxide thin films at low temperatures. The key mechanisms are explained in relation to the specific step of the fabrication process reached in an earlier stage: the formation of a defect-free, highly densified amorphous metal–oxygen network or the actual crystallization of the metal oxide. The role of photochemistry, where light can be used as a complementary energy source to induce crystallization, is particularly highlighted. This requires the synthesis of novel photosensitive solutions (modified metal alkoxides, charge-transfer metal complexes or structurally designed molecular compounds) and a precise control over the reactions promoted by UV irradiation (photochemical cleavage, ozonolysis, condensation or photocatalysis). Relevant examples derived from the integration of crystalline metal oxide thin films on flexible substrates (≤350 °C) illustrate the most recent achievements in this field.


 Please wait while we load your content...
Please wait while we load your content...