Rotational spectra and theoretical study of tetramers and trimers of 2-fluoroethanol: dramatic intermolecular compensation for intramolecular instability†
Abstract
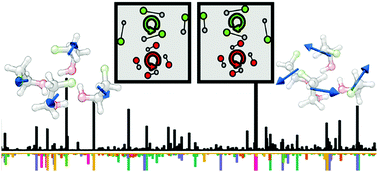
Using broadband rotational spectroscopy aided by high level ab initio calculations, we probe structural diversity and emerging bulk behavior in trimeric and tetrameric aggregates of the transiently chiral 2-fluoroethanol (FE). One FE tetramer which is homochiral and features a highly compact arrangement that is stabilized by a water tetramer-like H-bond ring and a network of fully bifurcated C–H⋯F H-bonds, and two higher energy FE trimers which feature a water trimer-like H-bond ring, were observed experimentally in a jet expansion. The three most stable FE trimers predicted within a few kJ mol−1 are all made of the most stable FE subunit and are detected experimentally. Unexpectedly, two out of the three most stable FE tetramers exclusively consist of much less stable subunits. For example, one FE tetramer that contains four less stable subunits, which are in total ∼40 kJ mol−1 less stable than their global minimum counterparts, is predicted to be of similar stability as the tetramer containing four global minimum subunits. High level theoretical modeling is essential in providing a comprehensive picture of the energetic and structural landscapes of the FE tetramer, an intriguing system at the interface between gas- and bulk-phase behavior, where the conformational specificity seen in the gas-phase is still experimentally relevant but plays a diminished role relative to the intermolecular topology and cooperative stability.


 Please wait while we load your content...
Please wait while we load your content...