Pressure-induced phase transition, metallization and superconductivity in ZrS2
Abstract
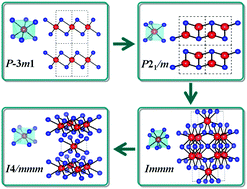
Zirconium disulfide (ZrS2) is an exemplary case among layered materials that exhibit unusual electronic and vibrational properties, with applications in potential photovoltaic and single-layer transistor materials. Here, we examine the effect of pressure on the structural stability, phonon dispersion, electronic properties and electron–phonon coupling of ZrS2 using first-principles calculations. Our results unravel that ZrS2 undergoes several pressure-induced phase transformations from the ambient-pressure P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 structure to a monoclinic P21/m structure at 2.0 GPa, to an orthorhombic Immm structure at 5.6 GPa, and to a tetragonal I4/mmm structure at 25.0 GPa. The electronic band calculations indicate that the layered P
m1 structure to a monoclinic P21/m structure at 2.0 GPa, to an orthorhombic Immm structure at 5.6 GPa, and to a tetragonal I4/mmm structure at 25.0 GPa. The electronic band calculations indicate that the layered P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 and P21/m structures are narrow-gap semiconductors. The gaps of the above two phases, which are normal semiconductors, decrease with pressure. Our results show that ZrS2 reaches the metallic state by a P21/m → Immm phase transition and keeps its metallic state in the I4/mmm phase. A pressure-driven evolution of the topological Fermi surface has been uncovered. The electron–phonon coupling results identify superconducting states in both metallic Immm and I4/mmm structures. Our research shows that pressure is efficient in the modulation of the bonding states, crystal structures and electronic properties of ZrS2, which will stimulate further high-pressure structural and conductive measurements.
m1 and P21/m structures are narrow-gap semiconductors. The gaps of the above two phases, which are normal semiconductors, decrease with pressure. Our results show that ZrS2 reaches the metallic state by a P21/m → Immm phase transition and keeps its metallic state in the I4/mmm phase. A pressure-driven evolution of the topological Fermi surface has been uncovered. The electron–phonon coupling results identify superconducting states in both metallic Immm and I4/mmm structures. Our research shows that pressure is efficient in the modulation of the bonding states, crystal structures and electronic properties of ZrS2, which will stimulate further high-pressure structural and conductive measurements.


 Please wait while we load your content...
Please wait while we load your content...