What's in a name?† ‘Coinage-metal’ non-covalent bonds and their definition
Abstract
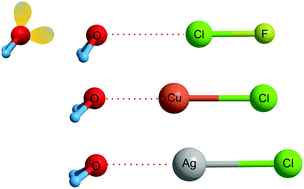
Many complexes of the type B⋯MX, (where B is a Lewis base such as H2, N2, ethyne, ethene, cyclopropane, H2O, H2S, PH3, or NH3, M is a coinage-metal atom Cu, Ag or Au, and X is a halogen atom) have now been characterised in the gas phase through their rotational spectra. It is pointed out that, for a given B, such complexes have angular geometries that are isomorphous with those of their hydrogen- and halogen-bonded counterparts B⋯HX and B⋯XY, respectively. Since the MX are, like the B, HX and XY referred to, closed-shell molecules, the complexes B⋯MX also involve a non-covalent bond. Therefore, the name ‘coinage-metal’ bond is suggested for the non-covalent interaction in B⋯MX, by analogy with hydrogen and halogen bonds. A generalised definition that covers all non-covalent bonds is also presented.
- This article is part of the themed collections: PCCP Perspectives and 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...