Cyanide–isocyanide isomerization: stability and bonding in noble gas inserted metal cyanides (metal = Cu, Ag, Au)†
Abstract
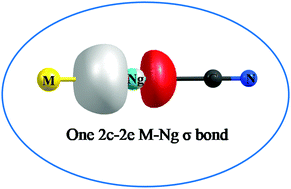
The internal isomerization, MNC ↔ MCN (M = Cu, Ag, Au), is investigated through quantum chemical computations. CuNC and AgNC are shown to be neither thermochemically nor kinetically stable against transformation to MCN. The free energy barrier (ΔG‡) for AuNC is somewhat considerable (7.1 kcal mol−1), indicating its viability, particularly at low temperature. Further, the Ng inserted analogues, MNgCN (M = Cu, Ag, Au; Ng = Xe, Rn) turn out to be thermochemically stable with respect to all possible dissociation channels but for two two-body dissociation channels, viz., MNgCN → Ng + MCN and MNgCN → Ng + MNC, which are connected to the internal isomerization processes, MNgCN → NgMCN and MNgCN → NgMNC, respectively. However, they are kinetically protected by substantial ΔG‡ values (11.8–15.4 kcal mol−1 for Cu, 9.8–13.6 kcal mol−1 for Ag, and 19.7–24.7 kcal mol−1 for Au). The pathways for such internal conversion are explored in detail. A thorough inspection of the bonding situation of the studied molecules, employing natural bond order, electron density, adaptive natural density partitioning, and energy decomposition analyses indicates that the M–Ng bonds in MNgCN and Ng–C bonds in AuNgCN can be represented as an electron-shared covalent bond. For the other Ng–C bonds, although an ionic description is better suited, the degree of covalent character is also substantial therein.


 Please wait while we load your content...
Please wait while we load your content...