Different scenarios of dynamic coupling in glassy colloidal mixtures†
Abstract
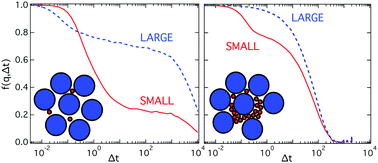
Colloidal mixtures represent a versatile model system to study transport in complex environments. They allow for a systematic variation of the control parameters, namely size ratio, total volume fraction and composition. We study the effects of these parameters on the dynamics of dense suspensions using molecular dynamics simulations and differential dynamic microscopy experiments. We investigate the motion of small particles through the matrix of large particles as well as the motion of large particles. A particular focus is on the coupling of the collective dynamics of small and large particles and on the different mechanisms leading to this coupling. For large size ratios, of about 1 : 5, and an increasing fraction of small particles, the dynamics of the two species become increasingly coupled and reflect the structure of the large particles. This is attributed to the dominant effect of the large particles on the motion of the small particles, which is mediated by the increasing crowding of the small particles. Furthermore, for moderate size ratios of about 1 : 3 and sufficiently high fractions of small particles, mixed cages are formed and hence the dynamics are also strongly coupled. Again, the coupling becomes weaker as the fraction of small particles is decreased. In this case, however, the collective intermediate scattering function of the small particles shows a logarithmic decay corresponding to a broad range of relaxation times.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...