Defect engineering in 1D Ti–W oxide nanotube arrays and their correlated photoelectrochemical performance†
Abstract
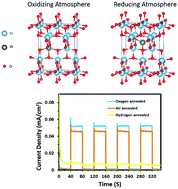
Understanding the nature of interfacial defects of materials is a critical undertaking for the design of high-performance hybrid electrodes for photocatalysis applications. Theoretical and computational endeavors to achieve this have touched boundaries far ahead of their experimental counterparts. However, to achieve any industrial benefit out of such studies, experimental validation needs to be systematically undertaken. In this sense, we present herein experimental insights into the synergistic relationship between the lattice position and oxidation state of tungsten ions inside a TiO2 lattice, and the respective nature of the created defect states. Consequently, a roadmap to tune the defect states in anodically-fabricated, ultrathin-walled W-doped TiO2 nanotubes is proposed. Annealing the nanotubes in different gas streams enabled the engineering of defects in such structures, as confirmed by XRD and XPS measurements. While annealing under hydrogen stream resulted in the formation of abundant Wn+ (n < 6) ions at the interstitial sites of the TiO2 lattice, oxygen- and air-annealing induced W6+ ions at substitutional sites. EIS and Mott–Schottky analyses indicated the formation of deep-natured trap states in the hydrogen-annealed samples, and predominantly shallow donating defect states in the oxygen- and air-annealed samples. Consequently, the photocatalytic performance of the latter was significantly higher than those of the hydrogen-annealed counterparts. Upon increasing the W content, photoelectrochemical performance deteriorated due to the formation of WO3 crystallites that hindered charge transfer through the photoanode, as evident from the structural and chemical characterization. To this end, this study validates the previous theoretical predictions on the detrimental effect of interstitial W ions. In addition, it sheds light on the importance of defect states and their nature for tuning the photoelectrochemical performance of the investigated materials.


 Please wait while we load your content...
Please wait while we load your content...