Mechanism of ionic-liquid-based acidic aqueous biphasic system formation†
Abstract
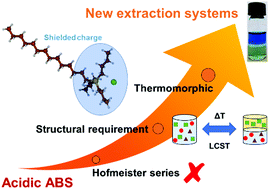
Ionic-liquid-based acidic aqueous biphasic systems (IL-based AcABS) represent a promising alternative to the solvent extraction process for the recovery of critical metals, in which the substitution of the inorganic salt by an acid allows for a ‘one-pot’ approach to the leaching and separation of metals. However, a more fundamental understanding of AcABS formation remains wanting. In this work, the formation mechanisms of AcABS are elucidated through a comparison with traditional aqueous biphasic systems (ABS). A large screening of AcABS formation with a wide range of IL identifies the charge shielding of the cation as the primary structural driver for the applicability of an IL in AcABS. Through a systematic study of tributyltetradecylphosphonium chloride ([P44414]Cl) with various chloride salts and acids, we observed the first significant deviation to the cationic Hofmeister series reported for IL-based ABS. Furthermore, the weaker than expected salting-out ability of H3O+ compared to Na+ is attributed to the greater interaction of H3O+ with the [P44414]+ micelle surface. Finally, the remarkable thermomorphic properties of [P44414]Cl based systems are investigated with a significant increase in the biphasic region induced by the increase in the temperature from 298 K to 323 K. These finding allows for the extension of ABS to new acidic systems and highlights their versatility and tunability.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...