The influence of aggregation on the third-order nonlinear optical property of π-conjugated chromophores: the case of cyanine dyes†
Abstract
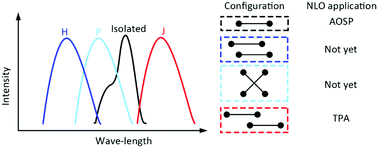
The external molecular environment like the aggregation of molecules can significantly change the intrinsic third-order nonlinear optical (NLO) property of π-conjugated chromophores. A combined experimental and theoretical study was performed to understand the influence of the aggregation of cyanines on the third-order NLO property in spin-coated thin films. Our result indicates that the H and J type cyanine dimers prefer the polyene-like structures and the P type dimer displays a comparatively smaller bond length alternation (BLA). The polarizable continuum model (PCM)-tuned, range-separated (RSE) density functional approach was used to describe the screening effect of the cyanine aggregation. In the thin film, the P aggregate has very small positive isotropic averaged second hyperpolarizability 〈γ〉, while the J aggregate has the largest positive 〈γ〉 due to the most polarized face-to-tail cyanine–cyanine interaction. Hence, the 〈γ〉 of the isolated cyanines (negative 〈γ〉) may get cancelled against that of the cyanine aggregates (positive 〈γ〉) in the thin film. The forward degenerate four-wave mixing technique also confirms a decrease in the magnitude of 〈γ〉 with an increase in the aggregation degree of cyanines. Since the large positive 〈γ〉 of the cyanine also implies strong two-photon absorption (TPA), the J aggregation of cyanines can be used as a potential fabrication method for applications involving TPA.


 Please wait while we load your content...
Please wait while we load your content...