Reaction kinetics of hydrogen atom abstraction from isopentanol by the H atom and HO2˙ radical†
Abstract
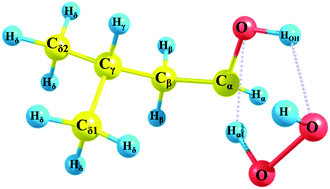
Isopentanol is a potential next-generation biofuel for future applications to Homogeneous Charge Compression Ignition (HCCI) engine concepts. To provide insights into the combustion behavior of isopentanol, especially to its auto-ignition behavior which is linked both to efficiency and pollutant formation in real combustion systems, detailed quantum chemical studies for crucial reactions are desired. H-Abstraction reaction rates from fuel molecules are key initiation steps for chain branching required for auto-ignition. In this study, rate constants are determined for the hydrogen atom abstraction reactions from isopentanol by the H atom and HO2˙ radical by implementing the CBS-QB3 composite method. For the treatment of the internal rotors, a Pitzer–Gwinn-like approximation is applied. On comparing the computed reaction energies, the highest exothermicity (ΔE = −46 kJ mol−1) is depicted for Hα abstraction by the H atom whereas the lowest endothermicity (ΔE = 29 kJ mol−1) is shown for the abstraction of Hα by the HO2˙ radical. The formation of hydrogen bonding is found to affect the kinetics of the H atom abstraction reactions by the HO2˙ radical. Further above 750 K, the calculated high pressure limit rate constants indicate that the total contribution from delta carbon sites (Cδ) is predominant for hydrogen atom abstraction by the H atom and HO2˙ radical.
- This article is part of the themed collection: Bunsentagung 2018: Kinetics in the Real World


 Please wait while we load your content...
Please wait while we load your content...