H-Abstraction reactions by OH, HO2, O, O2 and benzyl radical addition to O2 and their implications for kinetic modelling of toluene oxidation†
Abstract
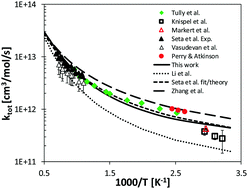
Alkylated aromatics constitute a significant fraction of the components commonly found in commercial fuels. Toluene is typically considered as a reference fuel. Together with n-heptane and iso-octane, it allows for realistic emulations of the behavior of real fuels by the means of surrogate mixture formulations. Moreover, it is a key precursor for the formation of poly-aromatic hydrocarbons, which are of relevance to understanding soot growth and oxidation mechanisms. In this study the POLIMI kinetic model is first updated based on the literature and on recent kinetic modelling studies of toluene pyrolysis and oxidation. Then, important reaction pathways are investigated by means of high-level theoretical methods, thereby advancing the present knowledge on toluene oxidation. H-Abstraction reactions by OH, HO2, O and O2, and the reactivity on the multi well benzyl-oxygen (C6H5CH2 + O2) potential energy surface (PES) were investigated using electronic structure calculations, transition state theory in its conventional, variational, and variable reaction coordinate forms (VRC-TST), and master equation calculations. Exploration of the effect on POLIMI model performance of literature rate constants and of the present calculations provides valuable guidelines for implementation of the new rate parameters in existing toluene kinetic models.
- This article is part of the themed collection: Bunsentagung 2018: Kinetics in the Real World


 Please wait while we load your content...
Please wait while we load your content...