In situ examination of a charged amino acid-induced structural change in lipid bilayers by sum frequency generation vibrational spectroscopy†
Abstract
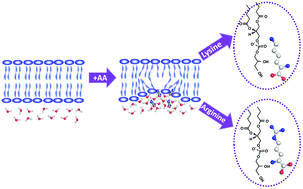
The interactions between amino acids (AAs) and membranes represent various short-range and long-range interactions for biological phenomena; however, they are still poorly understood. In this study, we used cationic lysine and arginine as AA models, and systematically investigated the interactions between charged AAs and lipid bilayers using sum frequency generation vibrational spectroscopy (SFG-VS) in situ and in real time. The AA-induced dynamic structural changes of the lipid bilayer were experimentally monitored using the spectral features of CD2, CD3, the lipid head phosphate, and carbonyl groups in real time. Time-dependent SFG changes in the structure of the lipid bilayer provide direct evidence for the different interactions of lysine and arginine with the membrane. It was found that the discrepancy between lysine and arginine in binding with the lipid bilayer is due to the nature of the terminal functional groups. Arginine exhibits a more drastic impact on the membrane than lysine. SFG responses of the acyl chains, phosphate groups, and carbonyl groups provide evidence that the interaction between AAs and the membrane most likely follows an electrostatics and hydrogen bond-induced defect model. This work presents an exemplary method for comprehensive investigations of interactions between membranes and other functionally significant substances.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...