Electrochemical surface-enhanced Raman spectroscopy (EC-SERS) study of the interaction between protein aggregates and biomimetic membranes†
Abstract
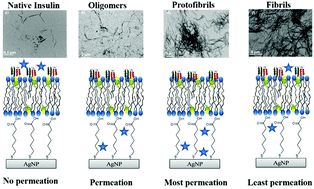
Human diseases characterized by the uncontrolled deposition of insoluble extracellular protein aggregates are collectively referred to as amyloidoses. Such diseases include Alzheimer's, Parkinson's, Huntington's, and prion disease. In Alzheimer's disease, it is believed that amyloid-β proteins may be responsible for pore and defect formation within cellular membranes, leading to a breakdown of cellular homeostasis causing eventual neuronal death. This theory is referred to as the amyloid pore hypothesis of Alzheimer's disease. In this work, the interaction between a model amyloid-forming protein (insulin) and a biomimetic membrane was studied at the molecular level. Protein at different stages of aggregation was allowed to interact with a biomimetic membrane formed on a nanostructured substrate using Langmuir–Blodgett/Langmuir–Schaefer deposition. Electrochemical surface-enhanced Raman spectroscopy (EC-SERS) was used to monitor the molecular level changes occurring as a result of this interaction. Based on the results it was observed that oligomers and protofibrils caused the most significant membrane deterioration whilst native protein appeared to play a protective role. To the best of our knowledge, this work represents the first EC-SERS investigation of protein aggregate–biomembrane interactions, and highlights the usefulness of this tool for studying complex biomolecular interactions.


 Please wait while we load your content...
Please wait while we load your content...