Dynamic wetting of imidazolium-based ionic liquids on gold and glass
Abstract
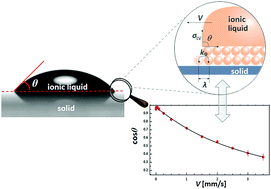
Many of the applications of ionic liquids rely on their bulk properties or their solvation abilities. However, it is their interactions with solid surfaces that underpin many of their potential applications in advanced technologies. Whether it is as lubricants for wind turbines or as electrolytes in supercapacitors, there are many areas where ionic liquids can provide an improvement in performance relative to more commonplace liquids. However, there are some barriers to their implementation in such applications. Foremost of these is the lack of systematic studies of their interactions with solid surfaces as well as neglecting the effect of the absorbed water on wetting. The present study explores the dynamic wetting of three ionic liquids (with a different length of hydrocarbon chain in the cation) on gold and glass substrates, both of which are relevant for nano- and micromechanical machine applications, under well-controlled environmental conditions. The form of data capture (Wilhelmy plate) allows for a direct analysis using analytical expressions for the two dominant approaches for dynamic wetting: the hydrodynamic and molecular kinetic models. All ionic liquids yield data that are described best by the molecular kinetic model. Substrate-ionic liquid and water–ionic liquid interactions contribute to the mechanisms involved in the wetting process.


 Please wait while we load your content...
Please wait while we load your content...