Exploring polymorphism and stoichiometric diversity in naproxen/proline cocrystals†
Abstract
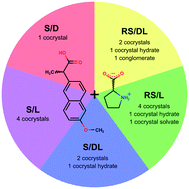
We present naproxen/proline cocrystals discovered when combining enantiopure and racemic naproxen and proline. Using liquid-assisted grinding as the main method to explore the variety of crystal forms in this system, we found 17 cocrystals, of which the structures of only four of them were previously known. The naproxen/proline system exhibited multiple polymorphs of 1 : 1 stoichiometry as well as more rare cocrystals with 1 : 2 and 2 : 3 stoichiometries, two cocrystal hydrates and one cocrystal solvate. In situ ball-milling, used to monitor liquid-assisted grinding reactions, revealed that the solvent dictates the reaction intermediates even if the final reaction product stays the same. Synchrotron X-ray diffraction data collected in situ upon heating allowed us to monitor directly the phase changes upon heating and gave access to pure diffraction patterns of several cocrystals, thus enabling their structure determination from powder X-ray diffraction data; this method also confirmed the formation of a conglomerate in the RS-naproxen/DL-proline system. Proline in cocrystals kept its ability to form charge-assisted head-to-tail N–H⋯O hydrogen bonds, typical of pure crystalline amino acids, thus increasing the percentage of strong charge-assisted interactions in the structure and consequently providing some of the cocrystals with higher melting points as compared to pure naproxen. The majority of drugs are chiral, and hence, these data are of importance to the pharmaceutical industry as they provide insight into the challenges of chiral cocrystallization.
- This article is part of the themed collection: Editor’s Collection: Polymorphism in Molecular Crystals


 Please wait while we load your content...
Please wait while we load your content...